 Safety of COVID-19 vaccination in adults with prior COVID-19 infection, December 2020 – May 2021 ( study in 6.5 million v-safe participants ).unvaccinated people, December 2020 – July 2021 ( 6,974,817 unique individuals, vaccinated and unvaccinated, were included, of whom 3.3 million received at least one dose of Pfizer-BioNTech, 2.4 million received at least 1 dose of Moderna and 331,282 people received one dose of Janssen ) Assessing the risk of non-COVID-19 mortality among vaccinated vs.Safety monitoring of Janssen/J&J vaccine up to April 2021 ( assessing VAERS reports during a time when nearly 8 million doses of Janssen had been administered ).Identifying the risk of rare severe allergic reactions among Moderna-vaccinated people, December 2020 – January 2021 ( 4.0 million doses of Moderna ).A broad assessment of COVID-19 vaccine safety, 2020 -2021 ( 4.1 million Pfizer-BioNTech vaccines, 2.6 Moderna vaccinees and 0.4 million Janssen vaccinees included ).An early assessment of vaccine reactogenicity in the 2 weeks following mRNA vaccination ( December 2020 – March 2021, including 4.7 million v-safe registrants ).vaccination program ( December 2020 – June 2021, involving 7.9 million v-safe participants) An early assessment of mRNA vaccine safety during the first 6 months of the U.S.Specific U.S.-based safety investigations have included (not an exhaustive list): VAERS, v-safe and VSD have all been continuously monitoring the safety of COVID-19 vaccines since they were originally rolled out. History of COVID-19 vaccine safety investigations in VAERS and VSD In addition to common and usually nonserious adverse effects such as injection site pain, fatigue and headache, some more serious safety signals have been identified in relation to COVID-19 vaccination thanks to a combination of the vaccine safety infrastructures described above. Until May 2023, CDC’s v-safe smartphone-based reporting system collected user-input data for COVID-19 vaccines specifically. VSD has been in existence since 1990 and currently includes electronic health data for approximately 3% of the U.S. These hypotheses may come from VAERS, v-safe or medical literature. Information from the electronic health records of individuals at these managed-care organizations is combined to investigate formally hypothesized relationships between vaccines and specific safety outcomes. VSD is composed of several large managed-care organizations in the U.S. Instead, it is an early warning system used to generate hypotheses about vaccine safety that can then be investigated using more robust methods. VAERS is not structured to formally investigate causal relationships between vaccines and potential safety outcomes ( Dusto, May 2022). Health care providers are required to report certain COVID-19 vaccine safety outcomes to VAERS, but anyone can report events to VAERS, including care providers, child care professionals and individual patients/laypeople. VAERS is an open database that collects reports of adverse events following vaccination. Vaccine safety infrastructure in the U.S. Each of these systems plays a distinct and important role in COVID-19 vaccine safety investigations in the U.S. 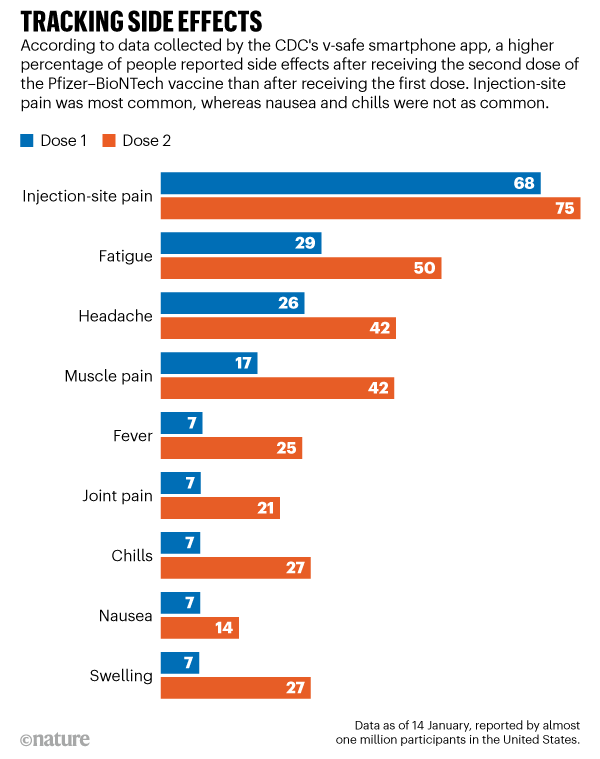
Additional safety investigations and considerations are performed by FDA’s BEST system and by using information from the Centers for Medicare & Medicaid Services. 
These include the Vaccine Adverse Event Reporting System ( VAERS) and the Vaccine Safety Datalink ( VSD). There are several infrastructures in place to investigate vaccine safety outcomes after vaccine authorization or licensure in the United States. Therefore, vaccines continue to be assessed for safety in much larger populations (millions or tens of millions of people) after they are authorized and/or licensed for use. Because background rates of these outcomes are very low, the only way to appropriately assess differences in risks of these outcomes is to measure relative risks in large populations. 
These could include outcomes such as Guillain-Barré syndrome, ischemic stroke or myocarditis. Sometimes, rare events may occur after vaccination that are too rare to be identified within clinical trials. Reactogenic events can include local outcomes such as injection site pain and redness/swelling at the injection site, as well as systemic outcomes including myalgia, arthralgia, fever and headache ( Hervé, September 2019). Vaccines are said to be reactogenic or have high reactogenicity if they elicit substantial inflammation subsequent to immunization. In clinical trials, vaccines are usually assessed for local and systemic events. The safety of vaccines is evaluated consistently from clinical trials all the way to post-authorization/post-licensure studies. How are vaccine safety questions answered and by whom? 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
